These configurations are anomalous, as one electron from the 4s orbital moves to the 3d orbital, leading to half-filled and fully-filled 3d orbitals. Chromium's electronic configuration is 3d^5 4s^1, and copper's electronic configuration is 3d^10 4s^1. Chromium (Cr) and Copper (Cu)Ĭhromium and copper are two elements that deviate from the Aufbau Principle. These exceptions occur when the energy difference between orbitals is small, and it is more energetically favorable for electrons to fill a higher energy orbital than to follow the Aufbau sequence. Electron configurations and orbital diagrams can be determined by applying the Pauli exclusion principle (no two electrons can have the same set of four. Exceptions to the Aufbau PrincipleĪlthough the Aufbau Principle provides a general guideline for the filling of atomic orbitals, there are some exceptions to the rule. The first shell, or energy level, is represented by the number 1, while the sublevel (s, p, d. creates bonds from overlap of atomic orbitals ( s, p, d ) and hybrid orbitals ( sp, sp2, sp3 ) combines atomic orbitals to form molecular orbitals (,, , ) forms or bonds. considers electrons delocalized throughout the entire molecule. 
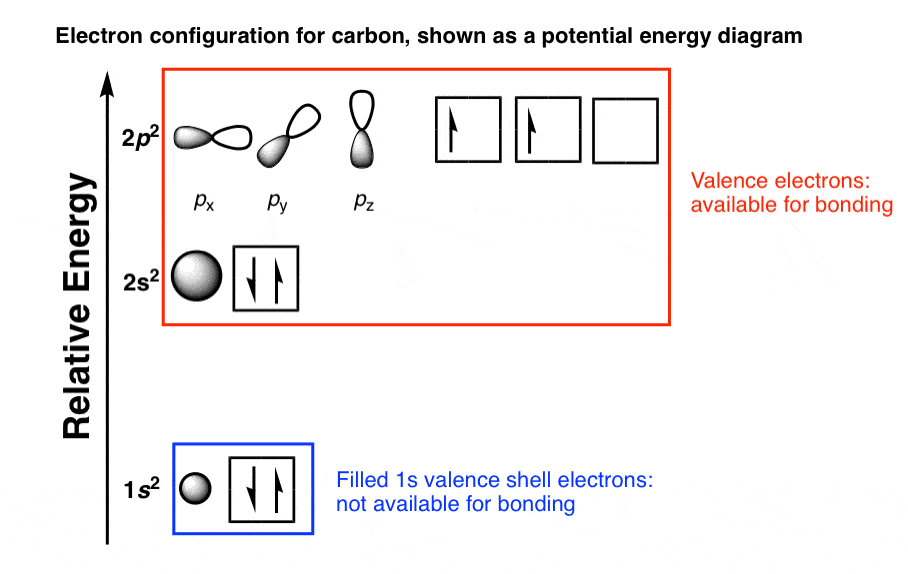
Elements are arranged in the periodic table based on their electronic configurations, with elements in the same group having similar configurations. For example, the electron configuration of carbon is 1s2 2s2 2p2. considers bonds as localized between one pair of atoms. The Aufbau Principle forms the basis for the organization of the periodic table of elements. The knowledge of the electronic configuration of atoms, obtained using the Aufbau Principle, is critical in interpreting electronic spectroscopy data. Electronic SpectroscopyĮlectronic spectroscopy is a technique used to study the electronic properties of atoms and molecules. The Aufbau Principle helps in predicting the types of chemical bonds that atoms form based on their electronic configurations. The knowledge of the electronic configuration of atoms is important in understanding chemical bonding. The Aufbau Principle finds applications in several areas of chemistry, such as: Chemical Bonding
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
